 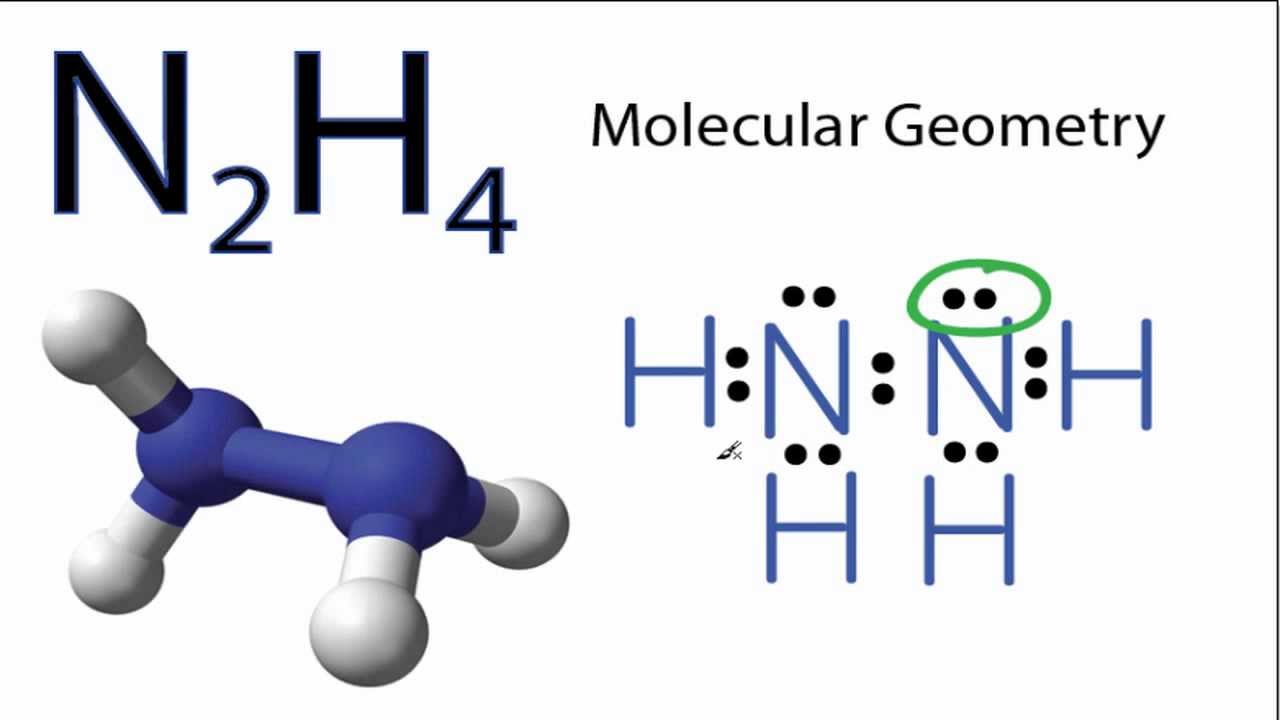 
In the following section, we will learn in detail about the geometric shape of this compound. The geometric structure of this compound is a trigonal pyramid in shape. So, the combination has a stable structure now. 
Now, if you check the surrounding electrons of both the compounds, you can see each Hydrogen atom has two surrounding atoms, while the phosphorous atom has eight electrons around it. The remaining two unpaired electrons of phosphorous are placed on the 4th side that forms a lone pair. Three valence electrons of phosphorous forms pairs with three valence electrons from the hydrogen atoms. Since hydrogen has one valence electron, and phosphorous has three, so P is the central atom in the molecular structure of this compound. The electronic configuration of the atoms let us know how many atoms can participate in the bonding. In Phosphene, three hydrogen atoms combine with phosphorous. It means hydrogen has one valence electron while phosphorous has three. If you look at the periodic table, you will see hydrogen is placed in the first column while phosphorous in the 5th column. Phosphorous has an electronic configuration of 1S2 2S2 2P6 3S2 3P3. Hydrogen has an electronic configuration of 1S1 as it has only one electron. To understand the structure of PH3, we should know the electronic configuration of the atoms and how many valence electrons are there in the atoms. The molecular formula of phosphene is PH3 which indicates the compound has one phosphorous atom bonding with three hydrogen atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
